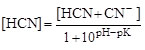Cyanide in freshwater and marine water
Toxicant default guideline values for protecting aquatic ecosystems
October 2000
Extracted from Section 8.3.7 ‘Detailed descriptions of chemicals' of the ANZECC & ARMCANZ (2000) guidelines.
The default guideline values (previously known as ‘trigger values’) and associated information in this technical brief should be used in accordance with the detailed guidance provided in the Australian and New Zealand Guidelines for Fresh and Marine Water Quality.
Description of chemical
Cyanides are organic and/or inorganic compounds which contain the cyano group –CN. Cyanide (CAS 151-50-8 when in the form of KCN) is a common industrial chemical used for nitrile and methacrylate fibres and other organic nitrile compounds; extraction of gold and silver from low grade ores, in electroplating and metal production (e.g. steel) (CCREM 1987). Cyanide also has other uses in pesticides (Leduc et al. 1982). It is a by-product of coke and gas production and can be found naturally in some plants such as bitter almonds, lima beans and cassava (CCREM 1987).
The cyanides present in effluents may be of different forms such as hydrocyanic acid HCN, cyanide ion CN-, various metallo-cyanide complexes which span a wide range of stabilities ([M(CN)n]m-), cyanogen (CN)2, cyanates (containing -OCN), thiocyanates (-SCN) and nitriles (RCN, R for alkyl group). Free cyanide is the sum of cyanide present as molecular HCN and ionic CN- whereas total cyanide includes also the measurable cyanide from breakdown of metallo-cyanide and organic complexes.
The different forms of cyanide have different chemical properties, and hence different degrees of toxicity to aquatic organisms. The HCN and CN- present or derived from dissociation of complexed or bound cyanides are the principal toxic forms (Doudoroff et al. 1966, Broderius et al. 1977), the former being more toxic because it is able to cross biological membranes. The toxicity of cyanides is mainly through the inhibition of cellular respiration. The binding of cyanide to haeme iron(III) of enzymes such as cytochrome oxidase, prevents electron transfer to molecular O2.
Environmental fate
The form of cyanide in water is affected by pH, temperature, dissolved oxygen, salinity, other ions, complexing agents and sunlight (Leduc et al. 1982, CCREM 1987). HCN is a readily diffusible, quite volatile and highly reactive substance. It is a weak acid in aqueous solution and the proportion of cyanide present as free cyanide depends on pH and temperature (CCREM 1987). Its dissociation in water is represented by:
HCN ≡ H+ + CN-
The relation of degree of dissociation to temperature is given by (Broderius et al. 1977):
where T is t°C. The concentration of un-ionised HCN in the free cyanide can be derived at various levels from:
where [HCN + CN-] is the free cyanide concentration. The fraction of un-ionised HCN at different pH and temperatures could be calculated using the above formulas. In most environmental situations at low temperature, most of the free cyanide comprises HCN. For instance, at 25°C and pH ≤ 8, when no other forms of cyanide are present, the fraction of HCN to the total free cyanide is at least 95%. Table 8.3.8 gives the proportion of un-ionised HCN to free cyanide (HCN + CN-) at pH 6.5 to 9.0 and temperatures between 10 and 30°C.
| Temp (°C) | pH | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 6.5 | 6.6 | 6.7 | 6.8 | 6.9 | 7.0 | 7.1 | 7.2 | 7.3 | 7.4 | 7.5 | 7.6 | 7.7 | 7.8 | 7.9 | 8.0 | 8.1 | 8.2 | 8.3 | 8.4 | 8.5 | 8.6 | 8.7 | 8.8 | 8.9 | 9.0 | |
| 10 | 99.9 | 99.9 | 99.9 | 99.8 | 99.8 | 99.7 | 99.6 | 99.5 | 99.41 | 99.3 | 99.1 | 98.8 | 98.5 | 98.2 | 97.7 | 97.1 | 96.4 | 95.5 | 94.4 | 93.1 | 91.4 | 89.4 | 87.0 | 84.2 | 80.9 | 77.1 |
| 12.5 | 99.9 | 99.9 | 99.8 | 99.8 | 99.7 | 99.7 | 99.6 | 99.5 | 99.3 | 99.2 | 99.0 | 98.7 | 98.4 | 97.9 | 97.4 | 97.8 | 96.0 | 95.0 | 93.8 | 92.3 | 90.4 | 88.3 | 85.7 | 82.6 | 79.0 | 75.0 |
| 15 | 99.9 | 99.9 | 99.8 | 99.8 | 99.7 | 99.6 | 99.5 | 99.4 | 99.3 | 99.1 | 98.8 | 98.5 | 98.2 | 97.7 | 97.1 | 96.4 | 96.5 | 94.4 | 93.0 | 91.4 | 89.4 | 87.0 | 84.2 | 80.9 | 77.0 | 72.7 |
| 17.5 | 99.9 | 99.8 | 99.8 | 99.7 | 99.7 | 99.6 | 99.5 | 99.3 | 99.2 | 99.0 | 98.7 | 98.4 | 97.9 | 97.4 | 96.8 | 96.0 | 95.0 | 93.7 | 92.3 | 90.4 | 88.3 | 85.7 | 82.6 | 79.0 | 75.0 | 70.4 |
| 20 | 99.9 | 99.8 | 99.8 | 99.7 | 99.6 | 99.5 | 99.4 | 99.3 | 99.1 | 98.8 | 98.5 | 98.2 | 97.7 | 97.1 | 96.4 | 95.5 | 94.4 | 93.1 | 91.4 | 89.4 | 87.0 | 84.2 | 80.9 | 77.1 | 72.8 | 68.0 |
| 22.5 | 99.8 | 99.8 | 99.7 | 99.7 | 99.6 | 99.5 | 99.3 | 99.2 | 99.0 | 98.7 | 98.4 | 98.0 | 97.4 | 96.8 | 96.0 | 95.0 | 93.8 | 92.3 | 90.5 | 88.3 | 85.7 | 82.7 | 79.1 | 75.1 | 70.5 | 65.5 |
| 25 | 99.8 | 99.8 | 99.7 | 99.6 | 99.5 | 99.4 | 99.3 | 99.1 | 98.8 | 98.5 | 98.2 | 97.7 | 97.1 | 96.4 | 95.6 | 94.5 | 93.1 | 91.5 | 89.5 | 87.2 | 84.4 | 81.1 | 77.3 | 73.0 | 68.2 | 63.1 |
| 27.5 | 99.8 | 99.7 | 99.7 | 99.6 | 99.5 | 99.4 | 99.2 | 99.0 | 98.7 | 98.4 | 98.0 | 97.5 | 96.8 | 96.0 | 95.1 | 93.9 | 92.4 | 90.6 | 88.5 | 85.9 | 82.9 | 79.4 | 75.4 | 70.9 | 65.9 | 60.5 |
| 30 | 99.8 | 99.7 | 99.6 | 99.5 | 99.4 | 99.3 | 99.1 | 98.9 | 98.6 | 98.2 | 97.8 | 97.2 | 96.5 | 95.6 | 94.6 | 93.2 | 91.6 | 89.7 | 87.4 | 84.6 | 81.4 | 77.6 | 73.4 | 68.6 | 63.5 | 58.0 |
Cyanide binds with various heavy metal ions forming metallo-cyanide complexes with varying degrees of stability. For instance, Pb(II), Zn(II) and Cd(II) cyanide complexes are unstable and therefore dissociate readily in aqueous solution, forming CN- and HCN which are of greater proportion than the complex ions themselves. Less dissociation occurs with more stable complexes of Ni(II), Cu(II) and Ag(I). The degree of dissociation increases with decreased complex concentration, decreased pH and decreasing complex stability. Toxicity to aquatic organisms may be due partly to the complex ions although they are much less toxic than HCN. Ferric- and ferro-cyanides which have wide industrial uses, are stable complexes but readily release cyanide when exposed to ultraviolet light. Thus, sunlight causes the mobilisation of free cyanide in waters containing iron-cyanide complexes.
Cyanogen (CN)2 and cyanohydrins RR'C(OH)CN decompose in water to release free cyanide and therefore are toxic. Cyanogen chloride (CNCl) is the chlorination/oxidation product of different cyanide forms, and this chemical is extremely toxic. The other forms of cyanide such as thiocyanate, cyanate and nitriles do not form free cyanides (except for thiocyanate in acidic media) and hence are much less toxic.
Volatilisation is a significant removal process for free cyanide at high concentrations but the processes at low concentrations are not well understood (CCREM 1987). Complexation and oxidation, as well as microbial breakdown may also be important processes for removal of free cyanide. Leduc et al. (1982) indicated that cyanides do not necessarily have a short residence time in the environment.
Analytical methods
It is important that water managers and regulators are able to distinguish between cyanide complexed with iron and that bound in less stable complexes, as well as between complexed cyanide and free cyanide (HCN + CN-) or un-ionised HCN. In addition, in reporting cyanide concentration, the sample pH and temperature must also be included.
The methods commonly used for measuring cyanides include colorimetry, titrimetry or by potentiometry using a cyanide-selective electrode. Any of these methods may be preceded by a preliminary treatment of alkaline chlorination or distillation (with or without ultraviolet irradiation). These treatments enable measurement of cyanides, which may include free cyanide, the readily dissociable complexed cyanides or even the almost nondissociable cyanides. The chlorination process is appropriate for measuring the more dissociable forms of cyanide of intermediate stability, giving the so-called ‘cyanides amenable to chlorination’. The weak acid dissociable (WAD) cyanide is determined by rigorous distillation of a slightly acidified sample solution with elimination of stable iron-cyanide complexes (removed by precipitation or avoidance of UV light). The WAD cyanide refers to free cyanide and acid-dissociable complexes. Ultraviolet treatment of a sample enables the breakdown, and hence when coupled with rigorous distillation, it enables the measurement of stable iron-cyanides along with potentially dissociable complexes. For measuring free cyanide, the most appropriate method is the use of cyanide-selective electrode in conjunction with careful control of sample pH.
Aquatic toxicology
For cyanide, 24-hour LC50 values were included. Toxicity was high to most species (< 1000 µg/L). The values given below are geometric means (expressed as un-ionised HCN as µg CN/L) for species taken from all screened data at reported pH and temperature.
Freshwater fish: 22 species, 24 to 96-hour LC50, 40 to 1200 µg/L. Seventeen species were < 470 µg/L. The most sensitive was Salmo salar (24-hour geometric mean LC50 40 µg/L), while species Oncorhynchus mykiss showed most sensitive individual 24 to 96-hour LC50 of < 100 µg/L.
Freshwater crustaceans: nine species; 24 to 96-hour LC50, 90 to 2200 µg/L. Most figures were 100 to 500 µg/L. Low outlying figures of 1 and 3 µg/L were reported for D. pulex (Cairns et al. (1978) reported for the highest temperature 25°C; while lower temperatures ≤ 20°C had much higher figures. Chronic NOEC (reproduction) for Moinodaphnia macleayi was 20 µg/L (Australian data).
Freshwater insects: four species, 96-hour LC50, 432 to 512 µg/L. An additional species Tanytarsus dissimilis (midge) had a 48-hour LC50 of 2490 µg/L.
Freshwater molluscs: eight species, 48 to 96-hour LC50, 1080 to 791,000 µg/L.
Other freshwater invertebrates; Oligochaete Aeolosoma headleyi, 48-hour LC50, 9000 to 160,000 µg/L (figures below 10°C were 9000 to 10,000 and at 15°C and above were ≥ 120,000 µg/L). Rotifer, Brachyonus calyciflorus, 24-hour LC50, 62,400 µg/L. Platyhelminthes, Dugesia tigrina, 96-hour LC50, 2100 µg/L. Hydra, Hydra viridissima, 6-day chronic NOEC (population growth) of 67 µg/L.
Marine fish: two species, 96-hour LC50, 70 to 109 µg/L.
Marine crustaceans: two species of shrimp, 48 to 96-hour LC50, 110 to 250 µg/L. An additional species Artemia salina had a 24-hour LC50 of 6970 µg/L.
Marine molluscs: one species, Mytilus edulis, 96-hour LC50, 36,000 µg/L.
Marine annelid: one species, Dinophilus gyrociliatus 96-hour LC50, 5940 to 7570 µg/L.
Marine diatom: one species, Nitzschia closterium 72-hour EC50 (growth) of 57 to 270 µg/L and NOEC of 10 to 31 µg/L.
Australian and New Zealand data
Water flea Moinodaphnia macleayi, 5-day NOEC (reproduction) of 20 µg/L. Hydra viridissima, 6-day NOEC (growth) of 67 µg/L. Marine fish, black bream, Acanthopagrus butcheri, 96-hour LC50 of 70 µg/L; Australian bass, Macquaria novemaculata, 96-h LC50 of 109 µg/L. Marine shrimp Penaeus monodon, 96-hour LC50, 110 µg/L. Some larval development figures were reported for the doughboy scallop Mimachlamys asperrima, 48-hour EC50 between 29 and 686 µg/L and NOEC between 5 and 40 µg/L, but these were not used.
Almost all of these data were unique and could not be readily compared with overseas data.
Factors that affect toxicology
The factors described in Environmental fate above affect toxicity of cyanide. Cairns et al. (1978) reviewed the effect of temperature on toxicity of cyanide. Temperature effects on algal toxicity were inconclusive. Toxicity to rotifers, snails and water fleas increased with an increase in temperature. For instance, the 48-hour LC50 for the snail Nitocris sp decreased from 13,600 µg/L at 5°C to 7000 µg/L at 25°C. Similar 2-fold increases were reported for D. magna,D. pulex and a rotifer. The increase in toxicity of cyanide at higher temperature was explained in part by increased metabolism of the organism at higher temperature (Cairns et al. 1978). In contrast, the oligochaete Aeolosoma headleyi, showed the opposite trend with 48-h LC50 values of 9000 to 10,000 at 10 and 5°C, compared to 120,000 µg/L at 15°C and 160,000 µg/L at 20 and 25°C.
Cairns et al. (1978) did not report any effect of temperature on cyanide toxicity to five species of fish but they did notice a variation with different species. Brown (1968) found that HCN was more toxic to O. mykiss fry at 3°C than at 13°C. Smith et al. (1978) examined the effects of temperature HCN toxicity to fathead minnow Pimephales promelas, collected as field stock close to the target temperature. As temperature decreased from 30°C to 20°C, the LC50s increased only slightly from 157 to 174 µg/L HCN, to reach a maximum tolerance (191 µg/L) at 15°C in October (USA), then decreased to 167 µg/L at 5°C. The trend for brook trout Salvelinus fontinalis was clearer with a steady increase in toxicity at lower temperatures. The 96-hour LC50 varied from 53 µg/L at 4°C to 143 µg/L at 18°C. Toxicity to several fish species was around 4 times higher (lower LC50) at 31.4°C than at 26.5°C (Sarkar 1990). Similarly, toxicity to several crustaceans was around two times higher above 31°C but temperature did not appear to affect toxicity to insects or molluscs under similar conditions.
No pronounced correlation was found between acute toxicity of cyanide to fish and alkalinity or hardness (USEPA 1985c). Similarly, no correlation was found with cyanide toxicity and pH ≤8.3 as the proportion of the more toxic form un-ionised HCN to the combined concentrations of HCN and CN- is high (≥87%) at temperatures 10 to 30°C.
Guideline
The trigger values were derived from screened acute data conducted at different pHs (6.5 to 8.6) and temperatures (5 to 30°C). All values were first converted to concentration as un-ionised HCN using the formulas given above with the reported pH and temperature. As mentioned above, water managers need to distinguish between cyanide complexed with iron and that bound in less stable complexes, as well as between complexed cyanide and free cyanide (HCN+CN-) or un-ionised HCN. The pH and temperature of water samples need to be measured. Table 8.3.8 or the formulas given above can be used to calculate the un-ionised HCN fraction from free cyanide concentration to compare with the trigger value.
A freshwater moderate reliability trigger value for un-ionised HCN of 7 µg CN/L was calculated using the statistical distribution method with 95% protection and an acute-to-chronic ratio (ACR) of 8.45.
Acute EC50 figures for D. pulex of 1 and 3 µg/L at 25°C were 100-fold lower than figures for D. pulex and, although it appears anomalous and requires further corroboration, it was used in the calculation to derive a geometric mean for this species.
A marine moderate reliability trigger value for un-ionised HCN of 4 µg CN/L was calculated using the statistical distribution method at 95% protection and an ACR of 8.45.
References
ANZECC & ARMCANZ 2000. Australian and New Zealand Guidelines for Fresh and Marine Water Quality, Australian and New Zealand Environment and Conservation Council and Agriculture and Resource Management Council of Australia and New Zealand, Canberra.
Broderius SJ, Smith LL & Lind DT 1977. Relative toxicity of free cyanide and dissolved sulphide forms to the fathead minnow. Canadian Fisheries Research Board Journal 36, 2323–2332.
Brown VM 1968. The calculation of the acute toxicity of mixtures of poisons to rainbow trout. Water Research 2, 723-733.
Cairns J Jr, Buikema AL Jr, Heath AG & Parker BC 1978. The effects of temperature on aquatic organism sensitivity to selected chemicals. Virginia Water Resources Research Center Bulletin 106, Blacksburg, Va.
CCREM 1987. Canadian water quality guidelines. Canadian Council of Resource and Environment Ministers, Ontario.
Doudoroff P, Leduc G & Schneider CR 1966. Acute toxicity to fish by solutions containing complex metal cyanides, in relation to concentrations of molecular hydrocyanic acid. Transactions of the American FisheriesSociety 95, 6–22.
Leduc G, Pierce RC & McCracken IR 1982. The effects of cyanides on aquatic organisms with emphasis upon freshwater fishes. National Research Council of Canada, Ottawa, NRCC 19246.
Sarkar SK 1990. Toxicity evaluation of sodium cyanide to fish and aquatic organisms: Effects of temperature. Science and Culture 54, 165–168.
Smith LL Jr, Broderius SJ, Oseid DM, Kimball GL & Koenst WM 1978. Acute toxicity of hydrogen cyanide to freshwater fishes. Archives of Environmental Contamination and Toxicology 7, 325-337.
USEPA 1985c. Ambient water quality criteria for cyanide — 1984. Criteria and Standards Division, US Environmental Protection Agency, Washington DC. EPA-440/5-84-028.